- Basic information
- Management and staff
- Ambulance
- Doctors
- Characteristics of the institute
- Department of Clinical Genetics
- Department of Medical Cytogenetics
- Department of Medical Molecular Genetics
- Center for Reproductive Genetics
- Department of Biology
- Overview of provided examinations and therapeutic procedures
- Documents to download
- Receipt of material
- Quality management
- Accreditation ČSN EN ISO 15189
- Participation in EQA
- Non-invasive prenatal testing (NIPT)
- FSGS
- ADPKD
- For the professional public
- For students
- National Coordinating Center for Rare Diseases
- About the project
- Project schedule
- Project description
- International cooperation
- Contact
- ACGT project
- EEA Funds 2014-2021
- Abbreviations
- Gallery
- Contact
Basic information
Department of Biology and Medical Genetics, 2nd Faculty of Medicine, Charles University and Motol University Hospital
ÚBLG has the character of a clinical-diagnostic, university and research workplace. The main goal is the diagnosis and study of hereditary diseases, including cancer at the gene and chromosomal level in the pre- and postnatal area. The staff are responsible for teaching biology, clinical and molecular genetics to Czech and foreign students of the 2nd Faculty of Medicine of Charles University in Prague. They also participate in the teaching of the University of the 3rd Age.
ÚBLG is a school-medical workplace. The school part is represented by the Department of Biology and Medical Genetics. The medical part consists of the Department of Clinical Genetics (outpatient part) and the laboratories: Department of Medical Cytogenetics and Department of Medical Molecular Genetics. The laboratory part is located on the 4th floor of node G (connecting part between the adult and children's part). The outpatient part of the institute is located in the adult part, node D, 3rd floor. The medical part is supplemented by the Center for Reproductive Genetics.

The head
Prof. MD Milan Macek jr., DrSc., MHA
(224) 433 500
milan.macek.jr@lfmotol.cuni.cz
Secretariat of the Motol University Hospital
Ing. Ivana Funková, MBA
(224) 433 500
ivana.funkova@fnmotol.cz
Secretariat of the 2nd Faculty of Medicine, Charles University
Bc. Alena Dimitriou
(224) 435 902
alena.dimitriou@lfmotol.cuni.cz
Head nurse
M.Sc. Hana Strouhalová
(224) 435 505
hana.strouhalova@fnmotol.cz

Medical Deputy Chief
Prim. MD Markéta Havlovicová
(224) 433 552
marketa.havlovicova@fnmotol.cz
Patient ordering
Working days 7:15-9:15 and 14:00-15:00
224 433 564, 224 433 567
More information on ordering patients can be found on a separate page.
Management and staff
Head of ÚBLG
Prof. MD Milan Macek Jr., DrSc., MHA
Email: milan.macek.jr@lfmotol.cuni.cz
tel .: (+420) 224 433 500-1
Medical Deputy Chief
MD Markéta Havlovicová
Email: marketa.havlovicova@fnmotol.cz
tel .: (+420) 224 433 552
Head of School
RNDr. Eduard Kočárek, Ph.D.
tel .: (+420) 257 296 156
Head nurse
M.Sc. Hana Strouhalová
Email: hana.strouhalova@fnmotol.cz
tel .: (+420) 224 433 505
Quality manager
RNDr. Tereza Stribna, Ph.D.
Email: tereza.stribna@fnmotol.cz
tel .: (+420) 224 433 539
Clinic manager
Ing. Ivana Funková, MBA
Email: ivana.funkova@fnmotol.cz
tel .: (+420) 224 433 500-1
Fax: (+420) 224 433 520
Secretariat
Ing. Ivana Funková, MBA (FN Motol)
Email: ivana.funkova@fnmotol.cz
tel .: (+420) 224 433 500-1
Fax: (+420) 224 433 520
Bc. Alena Dimitriou (2nd LF UK)
Email: alena.dimitriou@lfmotol.cuni.cz
tel .: (+420) 224 435 902
Head of ÚBLG:
- Department of Clinical Genetics - MD Markéta Havlovicová
- Department of Medical Cytogenetics - RNDr. Dear Novotná
- Department of Medical Molecular Genetics - MUDr. Anna Krepelova, CSc.
- Center for Reproductive Genetics - RNDr. Petra Paulasová, Ph.D.
- Department of Biology - Prof. Ing. Zdenek Sedlacek, DrSc.
Ambulance
ÚBLG offers general genetic counseling, specialized genetic counseling for cystic fibrosis and specialized genetic counseling for reproductive genetics. We take care of children and adult patients, or comprehensively for families.
Patient ordering
You can order a genetic consultation by phone at (+420) 224 433 564 and (+420) 224 433 567 on working days at 7:15-9:15 and 14:00-15:00. To order couples with fertility disorders, donors of reproductive cells, an embryological consultation or a spermiogram examination, call (+420) 224 433 578. We will also be happy to answer any professional questions at the e-mail address geneticka.ambulance@fnmotol.cz. Changes to the dates are also possible for our nurses at the numbers listed above, or at the e-mail address genetika.sestry@fnmotol.cz.
For general information on genetic testing, types of heredity, and some screening methods, see information materials, which arose within the project EuroGentest and were also translated into Czech. You can also read more detailed information about non-invasive prenatal testing (NIPT) in pregnancy.
Workplace location
Ambulance The Department of Clinical Genetics is located in the adult part of the main building of the Motol University Hospital, node D, 3rd floor.
Characteristics and focus of the workplace
The department focuses mainly on the following areas of disease: hereditary oncological diseases, neurogenetics - especially muscle diseases, ataxia, mental retardation and autism, chromosomal instability syndromes, microdeletion syndromes in neurology and cardiology, familial and syndromological growth disorders, genodermatoses, hearing disorders, rare cancers.
• Diagnosis of genetic diseases
• Dysmorphology
• Genetic prevention
Genetic clinic - accepts:
I. Patients in whose family
- an inherited disease or birth defect has occurred
- one or both partners suffer from an inherited or other chronic illness
- an affected child was born
- a marriage has occurred or is planned
- there were 2 or more miscarriages or sterility
- one or both partners were exposed to mutagenic influences (ionizing radiation, chemicals, drugs - eg cytostatics)
- one or both partners were treated for malignancy
- following positive neonatal screening
II. Pregnant women who
- were exposed to ionizing radiation (X-ray, CT)
- used drugs with a mutagenic or teratogenic effect
- underwent acute febrile illness in the first 3 months of pregnancy
- whose ultrasound or biochemical screening results are abnormal
- whose age is over 35 or the partner's age is over 45
Specialized genetic counseling for cystic fibrosis
- Molecular genetic diagnostics - prenatal and postnatal of all families at risk of cystic fibrosis.
- It reveals new mutations in the CFTR gene in typical and atypical forms of cystic fibrosis (CF).
- It provides early prevention of cystic fibrosis by examining CFTR gene mutations in partners with reproductive disorders, in children and adults with chronic diseases of the respiratory and gastrointestinal tract, in patients with tumors of the stomach, duodenum and pancreas.
Specialized genetic counseling for reproductive genetics
- Center for Reproductive Medicine and Reproductive Genetics in cooperation with the Gynecology and Obstetrics Clinic of the 2nd Faculty of Medicine of the UK and the Motol FN.
- Specialized genetic counseling services.
- Provision of other genetic examinations - clinical cytogenetics and molecular diagnostics in cooperation with the laboratory of clinical cytogenetics and molecular genetic laboratory of the national CF Center ÚBLG.
- Develops methods necessary for the clinical use of preimplantation genetic diagnostics.
As part of further genetic counseling of the workplace
- It ensures the detection of genetic predisposition to thromboembolic conditions by examining factor II mutations. (20210 A / G) and factor V. - Leiden in families and reproductive disorders, recurrent miscarriages, thrombosis of the cardiovascular system to prevent serious, life-threatening vascular events of the CNS and cardiovascular system. We are also investigating the 677 CT methyltetrahydrofolate reductase (MTHFR) gene polymorphism.
- Develops methods of molecular diagnostics of the most common aneuploidies of chromosomes 13, 18, 21, X and Y from microquantum cells for rapid prenatal and postnatal diagnosis of these aneuploidies and for the development of preimplantation diagnostics and prevention in cooperation with the Center for Reproductive Genetics and Medicine
- We offer pregnant women the opportunity to perform non-invasive prenatal testing (NIPT) - such as Panorama, Harmony and Prenatal Safe tests.
Doctors
The following doctors work at the Department of Clinical Genetics, ÚBLG FNM (sorted alphabetically by surname):
- MUDr. Miroslava Balascakova, Ph.D. - miroslava.balascakova@fnmotol.cz
- Prim. MUDr. Markéta Havlovicová - marketa.havlovicova@fnmotol.cz
- MUDr. Andrea Holubová - andrea.holubova@fnmotol.cz
- MUDr. Radka Kremlíková Pourová, Ph.D. - radka.pourova@fnmotol.cz
- MD Pavla Loníková — pavla.lonikova@fnmotol.cz
- MUDr. Marcela Malíková - marcela.malikova@fnmotol.cz
- MD Natalia Ptáčníková — natalie.ptacnikova@fnmotol.cz
- MUDr. Bc. Lukáš Ryba - lukas.ryba@fnmotol.cz
- MUDr. Pavel Tesner, Ph.D. - pavel.tesner@fnmotol.cz
- MUDr. Dana Thomas, Ph.D. - dana.thomasova@fnmotol.cz
- MUDr. Marek Turnovec - marek.turnovec@fnmotol.cz
- MUDr. Markéta Vlčková, Ph.D. - marketa.vlckova@fnmotol.cz
- MUDr. Emilie Vyhnálková, Ph.D. - emilie.vyhnalkova@fnmotol.cz
- MUDr. Veronika Zoubková - veronika.zoubkova@fnmotol.cz
Doctors currently on maternity / parental leave:
- MUDr. Nikola Čajbiková - nikola.cajbikova@fnmotol.cz
- MD Michaela Mihulová (Zelinová) — michaela.zelinova@fnmotol.cz
Characteristics of the institute
ÚBLG has the character of a clinical-diagnostic, university and research institution. The main goal of the institute is the diagnosis and study of hereditary diseases at the gene and chromosomal level in the prenatal and postnatal period.
ÚBLG is an educational and healthcare workplace. It represents the school part Department of Biology. The medical part consists of Department of Clinical Genetics (ambulatory part) and from individual laboratories: Department of Medical Molecular Genetics, Department of Medical Cytogenetics a Centers of Reproductive Genetics.
The eligibility of the ÚBLG is based on the following documents:
Statute of the University Hospital in Motol dated 20 July 7 and the Articles of Association of the University Hospital in Motol dated 2012 May 29 (File no. MZDR 5 2012-IV / 17).
Accreditation of ÚBLG Laboratories since 2009 according to the international standard ČSN EN ISO 15189 "Medical laboratories - Requirements for quality and competence" (ČIA).
The scope of the granted accreditation is on examination methods of molecular genetics, cytogenetics, laboratory andrology for IVF and primary sampling.
Accreditation of SAK FN Motol (Joint Accreditation Commission, ops). FN Motol successfully passed the investigation of the Joint Accreditation Commission and met the criteria for management and continuous quality improvement according to national accreditation standards. The medical facility holds a Certificate of Accreditation from the University Hospital in Motol (since 2011).
SÚKL decision to permit the operation of tissue establishments and diagnostic laboratories on the basis of inspections performed at the workplace of the Center for Reproductive Genetics, Department of Medical Molecular Genetics and Department of Medical Cytogenetics within the scope of authorization pursuant to Act No. 296/2008 Coll. intended for human use and amending related laws (Human Tissues and Cells Act).
Consent of the Ministry of Health to the implementation of procedures and methods of assisted reproduction (meeting no .: 25751/2006, date of issue: 7 September 9) in the Center for Reproductive Genetics within the Center for Reproductive Medicine and Reproductive Genetics.
Accreditation of the Ministry of Health of the Czech Republic for the implementation of the practical part of the specialization educational program in the field of "Medical Genetics" (Decision of the Ministry of Health of the Czech Republic
Accreditation of the Ministry of Health of the Czech Republic for the implementation of the practical part of the specialization educational program in the field of "Clinical Genetics" (Decision of the Ministry of Health of the Czech Republic
Accreditation of the Ministry of Health of the Czech Republic for the implementation of the practical part of the specialization educational program in the field of "Clinical Genetics" (for medical laboratory technicians) (Decision of the Ministry of Health of the Czech Republic of 9.8.2011 File no.
Department of Clinical Genetics
ÚBLG offers general genetic counseling, specialized genetic counseling for cystic fibrosis and specialized genetic counseling for reproductive genetics. We take care of children and adult patients, or comprehensively for families.
Patient ordering
You can order a genetic consultation by phone at (+420) 224 433 564 and (+420) 224 433 567 on weekdays from 7:15 a.m. to 9:15 a.m. and 14:00 p.m. to 15:00 p.m. To order couples with fertility disorders, donors of reproductive cells, an embryological consultation or a spermiogram examination, call (+420) 224 433 578. We will also be happy to answer any professional questions at the e-mail address geneticka.ambulance@fnmotol.cz. Changes to the dates are also possible for our nurses at the numbers listed above, or at the e-mail address genetika.sestry@fnmotol.cz.
For general information on genetic testing, types of heredity, and some screening methods, see information materials, which arose within the project EuroGentest and were also translated into Czech. You can also read more detailed information about non-invasive prenatal testing (NIPT) in pregnancy.
Workplace location
Ambulance The Department of Clinical Genetics is located in the adult part of the main building of the Motol University Hospital, node D, 3rd floor.

Characteristics and focus of the workplace
The department focuses mainly on the following areas of disease: hereditary oncological diseases, neurogenetics - especially muscle diseases, ataxia, mental retardation and autism, chromosomal instability syndromes, microdeletion syndromes in neurology and cardiology, familial and syndromological growth disorders, genodermatoses, hearing disorders, rare cancers.
• Diagnosis of genetic diseases
• Dysmorphology
• Genetic prevention
Genetic clinic - accepts:
I. Patients in whose family
- an inherited disease or birth defect has occurred
- one or both partners suffer from an inherited or other chronic illness
- an affected child was born
- a marriage has occurred or is planned
- there were 2 or more miscarriages or sterility
- one or both partners were exposed to mutagenic influences (ionizing radiation, chemicals, drugs - eg cytostatics)
- one or both partners were treated for malignancy
- following positive neonatal screening
II. Pregnant women who
- were exposed to ionizing radiation (X-ray, CT)
- used drugs with a mutagenic or teratogenic effect
- underwent acute febrile illness in the first 3 months of pregnancy
- whose ultrasound or biochemical screening results are abnormal
- whose age is over 35 or the partner's age is over 45
Specialized genetic counseling for cystic fibrosis
- Molecular genetic diagnostics - prenatal and postnatal of all families at risk of cystic fibrosis.
- It reveals new mutations in the CFTR gene in typical and atypical forms of cystic fibrosis (CF).
- It provides early prevention of cystic fibrosis by examining CFTR gene mutations in partners with reproductive disorders, in children and adults with chronic diseases of the respiratory and gastrointestinal tract, in patients with tumors of the stomach, duodenum and pancreas.
Specialized genetic counseling for reproductive genetics
- Center for Reproductive Medicine and Reproductive Genetics in cooperation with the Department of Gynecology and Obstetrics, 2nd Faculty of Medicine, Charles University and Motol University Hospital.
- Specialized genetic counseling services.
- Provision of other genetic examinations - clinical cytogenetics and molecular diagnostics in cooperation with the laboratory of clinical cytogenetics and molecular genetic laboratory of the national CF Center ÚBLG.
- Develops methods necessary for the clinical use of preimplantation genetic diagnostics.
As part of further genetic counseling of the workplace
- It ensures the detection of genetic predisposition to thromboembolic conditions by examining factor II mutations. (20210 A / G) and factor V. - Leiden in families and reproductive disorders, recurrent miscarriages, thrombosis of the cardiovascular system to prevent serious, life-threatening vascular events of the CNS and cardiovascular system. We are also investigating the 677 CT methyltetrahydrofolate reductase (MTHFR) gene polymorphism.
- Develops methods of molecular diagnostics of the most common aneuploidies of chromosomes 13, 18, 21, X and Y from microquantum cells for rapid prenatal and postnatal diagnosis of these aneuploidies and for the development of preimplantation diagnostics and prevention in cooperation with the Center for Reproductive Genetics and Medicine
- We offer pregnant women the opportunity to perform non-invasive prenatal testing (NIPT) - such as Panorama, Harmony and Prenatal Safe tests. See more information <a href="https://cdn.shopify.com/s/files/1/1932/8043/files/200721_ODSTOUPENI_BEZ_UDANI_DUVODU__EN.pdf?v=1595428404" data-gt-href-en="https://en.notsofunnyany.com/">here</a>.
Department of Medical Cytogenetics
The department provides comprehensive services in prenatal and postnatal diagnosis of chromosomal defects by classical and modern methods. We perform examinations of karyotype and chromosome breaks, including acquired aberrations induced by epoxy-di-butadiene. We have a wide range of probes for FISH within the whole genome. We perform the array CGH method on oligonucleotide chips not only postnatally but also prenatally in fetuses with a high risk of congenital malformations, including examination of native amniotic fluid and CVS (chorionic villi).
The department also operates as a training facility in culture, cytogenetic and molecular cytogenetic methods, including a CGH array for pre- and postgraduate teaching. We organize internships and offer individual training in pre-certification training in the field of medicine, respectively. clinical genetics.
Head of Department: (+420) 224 433 562 - RNDr. Dear Novotná
EVALUATION OF CHROMOSOME ABERACTS OBTAINED
- Evaluation of chromosome aberrations obtained (411,58 KB)
FISH EXAMINATION
- FISH (424,87 KB)
EXAMINATION OF CHROMOSOME DEVIATIONS BY ARRAY CGH METHOD
HUMAN CELL CARYOTYPE EXAMINATION
- HUMAN CELL CARYOTYPE (429,85 KB)
Department of Medical Molecular Genetics
Our department provides diagnostic services - performs molecular genetic diagnostics - for outpatient physicians in the field of medical genetics ÚBLG, for specialists of other institutes and departments of the Motol University Hospital and for extramural workplaces, especially in the field of medical genetics, from all over the Czech Republic.
We perform highly specialized molecular genetic diagnostics in patients with inherited diseases and in their families using a wide range of molecular genetics methods. We provide isolation of human genomic DNA from blood, tissues or cells after cultivation. We perform DNA analysis of selected genes, especially genes responsible for cystic fibrosis, syndromes associated with mental retardation, ataxia, dystonia, syndromes with epilepsy, muscular dystrophy and myotonia, hereditary predisposition to cancer, rasopathy, selected osteochondrodysplasia, disorders and a number of other rare genetic diseases. We perform diagnostic tests, in indicated cases prenatal and predictive testing. For most of the analyzed genes, we are the only center in the Czech Republic providing DNA diagnostics. Our department is involved in national and international interlaboratory quality control.
We participate in solving research projects at the national and international level. As part of research projects, we are introducing new molecular genetic methods, analysis of new genes, gaining new knowledge about the nature of genetic diseases, the frequency, causes and manifestations of genetic diseases in our population. These findings allow us to provide a more accurate interpretation of molecular genetic analyzes.
Employees of our department are also involved in undergraduate teaching of clinical genetics students in the 4th year of the 2nd Medical Faculty of Charles University, in postgraduate teaching of biomedicine students of the 2nd Medical Faculty of Charles University and in postgraduate specialization of doctors and non-physicians in the field of medical and clinical genetics.
Head of Department: (+420) 224 433 576 - MUDr. A. Krepelova, CSc.
Provided molecular genetic tests
CYSTIC FIBROSIS
- Cystic fibrosis (414,91 KB)
THROMBOPHILIC MARKERS
- Thrombophilic markers (432,26 KB)
ANEUPLOIDIA
- Aneuploidy and sex determination by QFPCR method (425,01 KB)
SKELETAL DYSPLASIA AND CRANIOSYNOSTOS
- Achondroplasia (285,03 KB)
- Apert's syndrome (268,31 KB)
- Crouzon's syndrome (267,74 KB)
- Diastopic dysplasia (259,95 KB)
- Hypochondroplasia (271,75 KB)
- Camptomatic dysplasia (258,44 KB)
- Craniofrontonasal dysplasia (CFND) (258,10 KB)
- Lerri-Weill syndrome (268,33 KB)
- Muenke's craniosynostosis (266,82 KB)
- Non-syndromic craniosynostosis (395,96 KB)
- Pffeiffer syndrome (269,06 KB)
- Saethre-Chotzen syndrome (388,40 KB)
- Thanatophoric dysplasia (271,35 KB)
- X-dominant chondrodysplasia punctata (392,23 KB)
MENTAL RETARDATION SYNDROMES
- Allan-Herndon-Dudley syndrome (271,78 KB)
- Angelmann syndrome (264,37 KB)
- ARX-linked mental retardation (390,12 KB)
- Cohen's syndrome (273,70 KB)
- GLUT1 deficit (257,13 KB)
- Lőwe's oculocerebrorenal syndrome (261,39 KB)
- MECP2-linked mental retardation (260,76 KB)
- Mental retardation and autism by the NGS-CID_v2 panel (418,46 KB)
- FRAXE mental retardation (Gen FMR2) (258,70 KB)
- Mental retardation and X-linked mental retardation (340,51 KB)
- Mowat-Wilson syndrome (Hirschsprung's disease) (406,56 KB)
- Norrie's disease (260,91 KB)
- PHTS syndrome (391,47 KB)
- Prader-Willi syndrome (255,72 KB)
- Subtelomeric conversions (255,98 KB)
- Fragile X chromosome syndrome (286,97 KB)
GENDER DEVELOPMENT DISORDERS
- Camptomatic dysplasia with gender development disorder (258,44 KB)
- Disorders of gender development (404,52 KB)
- Gender determination (273,01 KB)
- Androgen insensitivity syndrome - AIS (266,40 KB)
INHERITIVE ATAXIES
- Autosomal dominant spinocerebellar ataxia (AD SCA) 1, 2, 3, 6, 7 (base panel) by TP-PCR and fragment analysis and autosomal dominant spinocerebellar ataxia (AD SCA) 8, 12, 17, 28 and DRPLA (extended panel) by TP-PCR, fragment analysis, sequencing of exons 15 and 16 of the AFG3L2 gene (409,39 KB)
- Autosomal recessive Friedreich's ataxia by TP-PCR and fragment analysis (396,17 KB)
- Familial hemiplegic migraine 1 and 3 (403,92 KB)
- Hereditary cerebellar ataxia EA2 (CACNA1A) and SPG7 (409,18 KB)
- FXTAS syndrome by TP-PCR (286,97 KB)
SPINKLE MUSCLE ATROPHY AND RELATED SYNDROMES
- Spinal muscular atrophy (260,11 KB)
- Spinobulbar muscular atrophy - Kennedy disease (260,53 KB)
DEGENERATIVE DISEASES OF BASAL GANGLI
- Neurodegeneration with iron accumulation NBIA2A (INAD) (391,99 KB)
- Pantothenate kinase neurodegeneration (PKAN, Hallervorden-Spatz syndrome) (266,48 KB)
DYSTONIA
- Alternating childhood hemiplegia and early-onset dystonia-parkinsonism (DYT12) (390,09 KB)
- ARX - linked epilepsy / mental retardation / dystonia (Partington's syndrome) (390,12 KB)
- BFIS (Benign Familial Infantile Seizures) and DYT 10 (Episodic Kinesigenic Dyskinesia 1) (260,12 KB)
- Cervical dystonia (DYT 24 and DYT 25) (395,77 KB)
- Dopa-responsive dystonia (DYT5) (270,75 KB)
- Idiopathic torsional dystonia (DYT1) (387,64 KB)
- Mohr-Tranebjaerg syndrome (395,96 KB)
- Myoclonic dystonia (DYT11) (389,06 KB)
- Primary dystonia linked to the THAP1 gene (DYT6) (392,16 KB)
SYNDROMES WITH EPILEPSY
- EFMR (epilepsy and MR limited to females; Dravet-like) (260,65 KB)
- EIEE1 (early infantile epileptic encephalopathy-1)(259,05 KB)
- EIEE4 (early infantile epileptic encephalopathy-4) (274,15 KB)
- Dravet Syndrome (SMEI) (299,63 KB)
INHERITANT NEUROPATHY
- Hereditary neuropathy CMT1A / HNPP (260,36 KB)
MUSCLE DYSTROPHY AND MYOTONIA
- Congenital myotonia (262,53 KB)
- Duchenne / Becker type muscular dystrophy (262,09 KB)
- Myotonic dystrophies type 1 and 2 (420,62 KB)
RASOPATHY
- Diseases from the group of RASopathy (430,88 KB)
OTHER GENETIC SYNDROMES
- Aniridia (389,40 KB)
- Beckwith-Wiedemann syndrome (403,69 KB)
- Beckwith-Wiedemann syndrome - clinical questionnaire (completed in PC) (19,29 KB)
- Beckwith-Wiedemann syndrome - clinical questionnaire (manual) (378,63 KB)
- BPES syndrome (259,23 KB)
- Catel Manzke syndrome (406,44 KB)
- Currarino syndrome (257,09 KB)
- Ehler-Danlos syndrome (EDS), autism and obesity (520,40 KB)
- Ectrodactyly, ectodermal dysplasia and cleft (EEC3) (258,89 KB)
- Floating Harbor syndrome (259,32 KB)
- Hereditary and idiopathic pancreatitis (422,03 KB)
- Holoprosencephaly (266,74 KB)
- Hypophosphatasia (267,31 KB)
- CHARGE syndrome (271,90 KB)
- Kabuki syndrome (426,65 KB)
- Capillary arteriovenous malformation-1 (CMAVM1) and arteriovenous malformation-2 (CMAVM2) (409,82 KB)
- Milroy's disease (402,62 KB)
- Multiple CNS cavernomas (CCM1, CCM2, CCM3) (268,82 KB)
- Multilocus imprinting disorder (MLID) (415,52 KB)
- Nephrogenic type I (OMIM 304800) and type II (OMIM 125800) diabetes insipidus (410,95 KB)
- Nijmegen breakage syndrome, NBS1 (258,38 KB)
- Norrie's disease (385,22 KB)
- Oculocutaneous albinism (type 1A and 1B) (405,03 KB)
- NPHP1 gene-bound diseases (421,74 KB)
- DFNB1 locus-related hearing loss (268,56 KB)
- Propionic acidemia (389,65 KB)
- Say-Barber-Biesecker variant of Ohdo syndrome (255,90 KB)
- Shimke's immunosuppressive dysplasia (389,89 KB)
- Silver-Russell syndrome (395,68 KB)
- Smith - Lemli - Opitz Syndrome (DHCR7) (410,86 KB)
- Treacher-Collins syndrome (392,68 KB)
- Selected hereditary diseases by the Unicorn panel (424,44 KB)
- X-linked juvenile retinoschiza (391,02 KB)
HEREDITARY PREDISPOSITION TO TUMORS
- Birt-Hogg-Dubé syndrome (257,57 KB)
- Cowden's syndrome (391,47 KB)
- Gorlin's syndrome (259,28 KB)
- Hereditary diffuse gastric cancer (261,10 KB)
- Hereditary breast and ovarian cancer (340,47 KB)
- Hereditary non-polyposis carcinoma of the colon - Lynch syndrome (358,70 KB)
- HLRCC syndrome (259,89 KB)
- Juvenile polyposis (279,14 KB)
- Legion's syndrome (392,03 KB)
- Li-Fraumeni syndrome (261,92 KB)
- Type I neurofibromatosis (423,25 KB)
- Papillary carcinoma of the kidney (260,89 KB)
- Peutz-Jeghers syndrome (261,88 KB)
- RTPS1 (Rhabdoid tumor predisposition syndrome-1) (260,01 KB)
- Von Hippel-Lindau disease(269,54 KB)
MICRODELECTIVE SYNDROMES
- Microdeletion syndromes (520,40 KB)
Center for Reproductive Genetics
The Center for Reproductive Genetics is a part of the Center for Reproductive Medicine and Reproductive Genetics of the University Hospital in Motol and the 2nd Medical Faculty of Charles University, which was established in 1995. reproductive medicine), which, through their long-term cooperation, professional, personnel, methodological and unique equipment, provide comprehensive services to couples with reproductive disorders based on the latest advances in medical genetics and clinical medicine.
Within the Center for Reproductive Medicine and Reproductive Genetics, patients receive the best and most modern medical and genetic care. The gynecologists of the integrated "Center" are developing ultrasound and endoscopic methods, which are the basis for determining the correct diagnosis and for proposing further treatment.
Detailed sperm examinations (sperm concentration and motility, morphological evaluation of spermatozoa, molecular cytogenetic examination of aneuploidies in spermatozoa) and cooperation with the Urological Clinic of the University Hospital in Motol enable comprehensive diagnostics and therapeutic care, including surgeries on the epididymis and testes (where sterility is conditioned by azoospermia). - absence of sperm in the ejaculate).
Reproductive genetics focuses on specialized genetic counseling for couples with reproductive disorders and develops in direct contact with improving methods of assisted reproduction.
We focus on improving preconception genetic diagnostics using methods of molecular genetics, molecular cytogenetics and classical cytogenetics (in cooperation with ÚBLG departments), and preimplantation genetic diagnostics in cooperation with colleagues from the cooperating center of assisted reproduction Repromeda.
In the embryology laboratory, thanks to experience and state-of-the-art technical equipment, all the procedures and procedures of assisted reproduction known at present are performed at a high level.
The center also provides treatment with donated gametes and embryos where necessary. Cryopreservation of sperm, possibly, is also standard. testicular tissue in cancer patients before starting oncotherapy, which can lead to fertility problems. Methods leading to successful cryopreservation of oocytes and ovarian tissue are being developed. In cooperation with the Department of Gynecology and Obstetrics, its perinatology center and our biochemical laboratory within the ÚBLG, subsequent comprehensive care in pregnancy after successful conception of assisted reproduction methods, including childbirth and the puerperium, is also guaranteed.
The Center for Reproductive Genetics has the approval of the Ministry of Health within the Center for Reproductive Medicine and Reproductive Genetics for the implementation of procedures and methods of assisted reproduction (Ref. No. 25751/2006, date of issue: 7 September 9).
The Tissue Facility and its cooperating diagnostic laboratories were the State Institute for Drug Control (SÚKL), based on inspections performed within the scope of authorization pursuant to Act No. 296/2008 Coll., On ensuring the quality and safety of human tissues and cells intended for human use and on change related acts (Act on Human Tissues and Cells) and pursuant to Act No. 552/1991 Coll., on state control, a decision on permitting the operation of a tissue facility and diagnostic laboratory was submitted on 15 November 11.
The permit was issued to the University Hospital in Motol to the extent specified in this decision.
The Center for Reproductive Medicine and Reproductive Genetics has been granted accreditation by a training facility for assisted reproduction methods. At the same time, it operates as a training facility for pre- and postgraduate teaching (also within the IPVZ Prague).
Department of Biology
The department provides teaching of medical biology and genetics and clinical and molecular genetics for domestic and foreign students of the 2nd Faculty of Medicine, Charles University. The research activity is focused on the study of germline mutations in the p53 gene in families with a high incidence of cancer, on the characterization of molecular aberrations in childhood and adult tumors (oncogene rearrangements and expression, microsatellite instability, methylation), molecular cytogenetics with a focus on microdeletion syndromes, syndromology and genetic issues with a focus on neurological diseases, the study of molecular aspects of cell signaling, and the study of human genome evolution.
Overview of provided examinations and therapeutic procedures
In this section you will find a complete overview of examinations and therapeutic procedures performed at our workplace. However, there is also information on the method of collection, transport of the primary sample and response time.
Molecular genetic testing
CYSTIC FIBROSIS
THROMBOPHILIC MARKERS
ANEUPLOIDIA
SKELETAL DYSPLASIA AND CRANIOSYNOSTOS
- Achondroplasia
- Apert's syndrome
- Crouzon's syndrome
- Diastopic dysplasia
- Hypochondroplasia
- Camptomatic dysplasia
- Craniofrontonasal dysplasia (CFND)
- Lerri-Weill syndrome
- Muenke's craniosynostosis
- Non-syndromic craniosynostosis
- Pffeiffer syndrome
- Saethre-Chotzen syndrome
- TAR syndrome
- Thanatophoric dysplasia
- X-dominant chondrodysplasia punctata
MENTAL RETARDATION SYNDROMES
- Allan-Herndon-Dudley syndrome
- Angelmann syndrome
- ARX-linked mental retardation
- Cohen's syndrome
- GLUT1 deficit
- Lőwe's oculocerebrorenal syndrome
- MECP2-linked mental retardation
- Mental retardation and autism by the NGS-CID_v3 panel
- FRAXE mental retardation (Gen FMR2)
- Mental retardation and X-linked mental retardation
- Mowat-Wilson syndrome (Hirschsprung's disease)
- Norrie's disease
- PHTS syndrome
- Prader-Willi syndrome
- Subtelomeric conversions
- Fragile X chromosome syndrome
GENDER DEVELOPMENT DISORDERS
- Camptomatic dysplasia with gender development disorder
- Disorders of gender development
- Androgen insensitivity syndrome - AIS
INHERITIVE ATAXIES
- Autosomal dominant spinocerebellar ataxia (AD SCA) 1, 2, 3, 6, 7 (basic panel) and autosomal dominant spinocerebellar ataxia (AD SCA) 8, 12, 17, DRPLA and 27B (extended panel) by TP-PCR and fragmentation analysis
- Autosomal recessive Friedreich's ataxia by TP-PCR and fragment analysis
- Familial hemiplegic migraine 1 and 3
- Hereditary cerebellar ataxia EA2 (CACNA1A) and SPG7
- FXTAS syndrome by TP-PCR
- CANVAS spectrum disorder
SPINKLE MUSCLE ATROPHY AND RELATED SYNDROMES
DEGENERATIVE DISEASES OF BASAL GANGLI
- Neurodegeneration with iron accumulation NBIA2A (INAD)
- Pantothenate kinase neurodegeneration (PKAN, Hallervorden-Spatz syndrome)
DYSTONIA
- Alternating childhood hemiplegia and early-onset dystonia-parkinsonism (DYT12)
- ARX - linked epilepsy / mental retardation / dystonia (Partington's syndrome)
- BFIS (Benign Familial Infantile Seizures) and DYT 10 (Episodic Kinesigenic Dyskinesia 1)
- Cervical dystonia (DYT 24 and DYT 25)
- Dopa-responsive dystonia (DYT5)
- Idiopathic torsional dystonia (DYT1)
- Mohr-Tranebjaerg syndrome
- Myoclonic dystonia (DYT11)
- Isolated dystonia linked to the THAP1 gene (DYT6)
SYNDROMES WITH EPILEPSY
- EFMR (epilepsy and MR limited to females; Dravet-like)
- EIEE1 (early infantile epileptic encephalopathy-1)
- EIEE4 (early infantile epileptic encephalopathy-4)
- Dravet Syndrome (SMEI)
INHERITANT NEUROPATHY
MUSCLE DYSTROPHY AND MYOTONIA
RASOPATHY
OTHER GENETIC SYNDROMES
- Aniridia
- Beckwith-Wiedemann syndrome
- BPES syndrome
- Catel Manzke syndrome
- Currarino syndrome
- Ehler-Danlos syndrome (EDS), autism and obesity
- Ectrodactyly, ectodermal dysplasia and cleft (EEC3)
- Floating Harbor syndrome
- Hereditary and idiopathic pancreatitis
- Holoprosencephaly
- Hypophosphatasia
- CHARGE syndrome
- Kabuki syndrome
- Capillary arteriovenous malformation-1 (CMAVM1) and arteriovenous malformation-2 (CMAVM2)
- Lymphatic malformations 4
- Multiple CNS cavernomas (CCM1, CCM2, CCM3)
- Multilocus imprinting disorder (MLID)
- Nephrogenic type I (OMIM 304800) and type II (OMIM 125800) diabetes insipidus
- Nijmegen breakage syndrome, NBS1
- Norrie's disease
- NPHP1 gene-bound diseases
- DFNB1 locus-related hearing loss
- Propionic acidemia
- Say-Barber-Biesecker variant of Ohdo syndrome
- Shimke's immunosuppressive dysplasia
- Silver-Russell syndrome
- Smith - Lemli - Opitz Syndrome (DHCR7)
- Treacher-Collins syndrome
- Selected hereditary diseases by the Unicorn panel
- X-linked juvenile retinoschiza
HEREDITARY PREDISPOSITION TO TUMORS
- Birt-Hogg-Dubé syndrome
- Cowden's syndrome
- Gorlin's syndrome
- Hereditary diffuse gastric cancer
- Hereditary breast and ovarian cancer
- Hereditary non-polyposis carcinoma of the colon - Lynch syndrome
- HLRCC syndrome
- Juvenile polyposis
- Legion's syndrome
- Li-Fraumeni syndrome
- Type I neurofibromatosis
- Papillary carcinoma of the kidney
- Peutz-Jeghers syndrome
- RTPS1 (Rhabdoid tumor predisposition syndrome-1)
- Von Hippel-Lindau disease
PHARMACOGENETICS
MICRODELECTIVE SYNDROMES
Cytogenetic examinations
EVALUATION OF CHROMOSOME ABERACTS OBTAINED
FISH EXAMINATION
- Microdeletion syndromes common and rare
- the so-called CATCH22 association, today referred to as the 22q11 chromosome microdeletion syndrome (this group includes DiGeorge's, velocardiofacial, or Shprintzen's syndrome)
- Prader-Willi / Angelman syndrome - microdeletion of chromosome 15q11-q13
- Williams-Beuren syndrome - microdeletion of chromosome 7q11.23
- Miller-Dieker syndrome - chromosome microdeletion 17p13.3
- Smith-Magenis syndrome - chromosome microdeletion 17p11.2
- microdeletion of TP53 gene chromosome 17p13.1
- Kallman syndrome - Xp22 chromosome microdeletion
- X-linked ichthyosis - STS (steroid sulfatase gene) locus microdeletion at Xp22
- SHOX gene deletion, duplication and translocation (Xp22.33; Yp11.3)
- microdeletion and translocation of the SRY region
- rare whole genome microdeletions / duplications
- Deletions and rearrangements of subtelomeric regions of chromosomes
- Chromosome marker structure
- Mosaicism
INVESTIGATION OF CHROMOSOMAL DEVIATIONS BY ARRAY CGH and SNP ARRAY
HUMAN CELL CARYOTYPE EXAMINATION
Methods of the Center for Reproductive Genetics
Laboratory examination in assisted reproduction techniques
Therapeutic procedures within assisted reproduction techniques
Biochemical examinations
Documents to download
Laboratory manual
Requests (indications) and clinical questionnaires for examination at our workplace
- Request for molecular genetic and cytogenetic examination (for completion in PC)
- Request for molecular genetic and cytogenetic examination (for manual completion)
- Spermiogram examination request (for completion in PC)
- Spermiogram examination request (for manual completion)
- Application for biochemical screening of congenital malformations with a clinical questionnaire
- Indications for prenatal examination
- Indications for treatment by AR methods
- Clinical Questionnaire - Beckwith-Wiedemann Syndrome (PDF)
- Clinical questionnaire - Beckwith-Wiedemann syndrome (MS Word)
- Clinical questionnaire - Intellectual insufficiency and autism
- Clinical questionnaire - Kabuki syndrome
- Clinical questionnaire - RASopathy (142,70 KB)
Tool for filling out a request form for a molecular-genetic or cytogenetic examination (see https://vysetreni.ublg.cz).
Informed patient consents / disagreements
- Genetic laboratory examination
- Prenatal genetic laboratory examination
- Prenatal screening for the most common chromosomal defects and some other fetal developmental defects
- Non-invasive prenatal screening (NIPT)
- Laboratory procedures of in vitro fertilization
- Comprehensive examination of sperm
- Cryopreservation of male reproductive cells
- Donation of male reproductive cells
Statement for treatment with assisted reproduction methods
Information for patients on the performed tasks
- What is anmiocentesis?
- Information and implementation of amniocentesis
- Genetic testing for health purposes
- Information on cystic fibrosis
- Patient information on genetic testing
- Information for the patient about invasive prenatal genetic testing
- Information leaflet
- Non-invasive prenatal testing
- Semen collection
- Instructions before collecting semen
Questionnaires for new patients
- Questionnaire - one patient
- Questionnaire - child and parents
- Questionnaire - couple (pregnant + partner/husband, couple planning pregnancy)
- Questionnaire - child caught by CF newborn screening
Pedigree questionnaire
Receipt of material
Central sample reception (Molecular genetic, cytogenetic examinations and samples for the examination of congenital malformations - biochemical screening):
FN Motol
V Úvalu 84
150 06, Prague 5
Building 3, Node G, 4th floor
Phone: +420 22443 3561, +420 22443 3581
Hours of Operation:
Working days: Mon - Fri
Working hours: 7:30 - 14.30:XNUMX
Tube mail: 3581, 3569, 3571
Receipt of samples (ejaculate) of the Center for Reproductive Genetics:
FN Motol
V Úvalu 84
150 06, Prague 5
New building, node D, 3rd floor, door no. 30071 (Room no. 9 CRG patient reception)
Phone: +420 22443 3578
Hours of Operation:
Working days: Mon - Fri
Working hours: 7:00 - 14:30 h
Tube mail: 3547
Quality management
The primary goal of the ÚBLG Laboratories is:
- provide contributing genetic tests to determine the diagnosis, or to clarify the diagnosis of the examined patients in accordance with Act 373/2011 Coll., on specific health services, Part 6 (as amended);
- provide genetic testing to determine the diagnosis, or to clarify the diagnosis of the examined patients in accordance with the Recommendations of the Society of Medical Genetics of the Czech Medical Association JEP, which reflect the recommendations of international professional societies and research consortia of the European Union;
- customer satisfaction ensuring a high standard of services providedwhich is given primarily maximum reliability of results performed tests and use contemporary investigative methods, which are in line with clinical needs and evidence-based medicine;
- constantly to develop qualifications and professional competence workers ;,
- effectively lead a functional quality management system, which meets the requirements of the international standard ČSN EN ISO 15189, Medical laboratories - Requirements for quality and competence (as amended);
- regularly set and review quality objectives. Familiarize all employees with the goals and results of the quality management system review;
- in the tissue facility of the Center for Reproductive Genetics meet the requirements of Act No. 296/2008 Coll., on ensuring the quality and safety of human tissues and cells intended for human use and requirements for the examination of donors to assess their medical fitness pursuant to Decree No. 422/2008 Coll .;
- ensure multi-source funding for the modernization and renewal of instrumentation to ensure the suitability of laboratory tests for the purpose and to continuously improve the quality of the laboratory services provided;
The aim of the quality management system is:
- adhere to processes before laboratory examination, ie to review the quality of delivered samples, their unambiguous identification, appropriate sample preparation in order to comply with the standard quality of laboratory testing
- secure high level of reliability of laboratory examination processes, using validated examination procedures, performing internal and external quality controls and performing regular maintenance of laboratory equipment;
- adhere to the specified sample response times;
- secure comprehensibility and clarity levels of findings issued by the laboratory thus, abThe information provided could be at most usable and understandable;
- to improve two-way communication laboratory staff with physician customers to ensure their satisfaction with the services provided;
- evaluate impacts of work processes and potential defects on the results of laboratory tests (risk management);
- monitor and evaluate laboratory processes in order to ensure efficiency their implementation and management;
- due to the potential risk of work in the processing of biological materials, pay due attention to all activities in the laboratory compliance with occupational safety and environmental protection;
Laboratory management is committed to meet the requirements of the standard ČSN EN ISO 15 189.
Prof. MD Milan Macek Jr., DrSc. Head of ÚBLG Laboratories Head of ÚBLG 2. LF UK and FN Motol
|
Date of approval: 15 December 12 |
Accreditation ČSN EN ISO 15189
The laboratories of ÚBLG, the Institute of Biology and Medical Genetics of the 2nd Medical Faculty of Charles University and the Motol University Hospital, are accredited by the Czech Accreditation Institute, ops (ČIA) based on the assessment of compliance with the accreditation criteria given by ČSN EN ISO 15 189.
The subject of accreditation are examination methods of molecular genetics, cytogenetics, laboratory andrology for IVF and primary sampling.
Accreditation certificate
- Accreditation certificate_EN (496,44 KB)
- Accreditation certificate_CZ - appendix (996,70 KB)
Certificate of Accreditation
- Certificate of accreditation_ENG (454,37 KB)
- Certificate of accreditation_ENG - appendix (971,76 KB)
Participation in EQA
Our workplace regularly successfully participates in interlaboratory external quality controls. Current certificates of our participation can be downloaded here.
- Cytogenetic examination - GenQA 2021 - Amniotic fluid, Blood - postnatal
- Cystic fibrosis - EQA CF Network 2022
- Examination of the most common aneuploidies by QF-PCR 2023
- Examination of thrombophilic markers - ÚHKT 33th cycle / May 2023
- Diagnosis of congenital malformations - SEKK VVV2 / 2022
- Diagnosis of congenital developmental defects - RfB SD1/2023
- Certificate of Participation - EMQN 2023
- DNA Sequencing - Sanger (ILR) EMQN 2023
- Sanger Sequencing - Method - RfB SQ2 / 23 Certificate
- Sanger Sequencing - Method - RfB SQ1 / 23 Certificate
- GenQA Clinical Genetics - Dysmorphology 2022
- GenQA Prenatal karyotyping 2022
Non-invasive prenatal testing (NIPT)
- Screening examination that detects the fetus with high accuracy especially Down syndrome from pregnant blood, without the need for invasive sampling (eg amniotic fluid collection, AMC): it can capture more than 99% of such fetuses.
- Health insurance this examination does not paybut some on it a certain amount contribute (VZP, ZPMV).
- The test is especially suitable for women with an increased age or biochemical risk of Down syndrome in the fetus, who do not wish to undergo an invasive examination, but also for low-risk women who wish to further reduce the risk of Down syndrome.
- Pre-subscription genetic consultation with a doctor - clinical geneticist.
- Testing is not a full-fledged replacement for invasive testing: not recommended if a fetal developmental defect is detected - in case of a defect finding, invasive examination may be recommended even after a favorable NIPT result; furthermore, it fails to elucidate other genetic burdens (eg monogenic diseases).
- Up to 3% of examinations require re-sampling, for some women it will not even allow to deliver the result (up to 1% of all examinations) - re-sampling is not paid by the pregnant woman;
- The need for re-sampling, test failure and a false negative result are more likely, especially in overweight / obese women, myomatosis, taking blood clotting drugs (eg Fraxiparine, Clexane, Anopyrin…).
- Failure of the test is a reason to recommend an invasive examination.
- Due to the possible false positivity of testing (approximately 0,1%), it is necessary to verify the positive result by an invasive method, most often AMC - in about 90% of these cases, Down's syndrome is confirmed; wider testing (microdeletion syndromes) significantly increases false positivity.
- Examination can rarely yield unexpected and unclear findings - for example, to detect genetic abnormalities or cancer in a pregnant woman.
- Examination is not possible in women after transplantation (especially bone marrow), up to 6 months after blood transfusion and in women with chromosomal abnormalities.
- In addition to Down's syndrome, the tests also detect rarer Edwards and Patau syndromes and allow the assessment of sex chromosome numbers (and fetal sex): the efficiency of detection of these other defects is lower (90-98%), the accuracy of sex determination is greater than 99%.
- Testing is possible from the completed 9-10. weeks, but is strongly recommended only after combined first trimester screening; with early testing, the pregnant woman will not be informed of the sex of the fetus before the completed 12th week (or screening of the first trimester) - with the exception of carriers of X-linked diseases.
- Testing is not recommended after the end of the 20th week.
- The results are available approximately 10 days after collection according to the test.
- Testing is possible in pregnancies after IVF - but this information is necessary for proper test evaluation, as well as other anamnestic information.
- When testing twin pregnancies, some test options are not available, the test parameters are slightly worse for twins; testing in triplets is not possible.
Further information will be provided to you by the attending physician of the genetic clinic, for more detailed questions please contact us by email pavel.tesner@fnmotol.cz.
It is possible to order by phone on working days between 7.15-8.00 and 14.00-15.00 from our nurses at 224 433 564 and 224 433 567.
At FN Motol, we currently offer you the option of taking the following tests (including other variants):
- Panorama (CZK 12 for the basic version of the test) - more at www.panoramatest.cz
- PrenatalSafe (CZK 12 for the basic test variant) - more at www.prenatalsafe.cz
- MaterniT21 + (CZK 12 and CZK 600) - more at www.materniT21.cz
FSGS
Do you have a diagnosis of focal segmental glomerulosclerosis (FSGS)?
Some patients have this diagnosis based on a mutation in one of the genes associated with kidney disease.
If your nephrologist does not find another reason for your diagnosis (for example, taking certain medications, certain types of infections, diabetes, high blood pressure, obesity, etc.), it is advisable to supplement the genetic test.
This examination can be crucial in caring for you and your relatives.
Ordered
You can order a genetic test by e-mail or telephone:
Email: michaela.zelinova@fnmotol.cz
Telephone: 601 082 807 (call on Friday, 8: 30-11: 00)
Frequently asked questions about genetic testing
Can I have a genetic disease when my problems only became apparent in adulthood? After all, genetic disease manifests itself in children.
Genetic disease can also manifest in adult patients. In the case of FSGS in adulthood, the mutation did not fully manifest itself until adulthood, but the predisposition to this disease was always present.
I'm the only one in the family, if it was a genetic / hereditary disease, then it would occur in someone else's family.
Even if you are the only patient in the family, it can be a genetic disease. The mutation could have arisen anew (you are the first carrier and there is a risk of transmission to your offspring). Or it is a recessive mutation, which means that your parents are carriers, and your children or siblings can be carriers or even disabled. That is why your examination is important not only for you, but also for your relatives.
Why is genetic testing important to me?
Patients with the genetic form of FSGS are known to respond differently to treatment than patients with the non-genetic form of the disease. Therefore, the result of the tests may change / streamline the treatment of your disease. Even in the case of kidney transplant patients.
As already mentioned, the results of your tests can help to detect in the family of another carrier of the mutation and determine whether he is not at risk of disease or whether he is not a carrier and there is no risk to his offspring.
What does genetic testing entail?
The genetic test is performed from a blood test tube. The test results last several months, because several tens to hundreds of genes are tested at once.
Ideally, the genetic examination also includes a consultation with a clinical geneticist, who will acquaint you with genetic testing and examine you clinically. This test can reveal signs typical of a particular gene's damage and speed up otherwise very long laboratory testing. The clinical geneticist may suggest additional medical tests to detect possible associated defects or offer other genetic testing based on family history.
ADPKD
Do you have a diagnosis of autosomal dominant polycystic kidney disease (ADPKD)?
It is a congenital disease based on a mutation in one of the genes associated with the proper functioning of the kidneys. Most often it is a gene PKD1 or PKD2, rarely other genes. We recommend genetic testing for patients / families with ADPKD for several reasons:
- Detection of the mutation will enable a possible prediction of the development of the disease (the course may differ depending on the affected gene but also the type of mutation).
- The disease often manifests itself in adulthood, genetic testing in the family will help reveal which family member inherited the talent and is at risk before the onset of clinical difficulties.
- If a mutation is found, couples can use the services of an assisted reproduction center and undergo artificial insemination with preimplantation embryo examination. In the case of spontaneous pregnancy, the risk of transmitting the disease to the offspring is up to 50%. Preimplantation examination allows you to select an embryo that does not have a talent. However, this artificial insemination must be preceded by genetic testing and detection of the mutation responsible for ADPKD.
Ordering for a genetic consultation / examination at the Motol University Hospital can be made by phone at 224 433 564 and 224 433 567 on working days at 7.15-8.00 and 14.00-15.00. It is also possible to order by email (this email can also be used in case of further questions): michaela.zelinova@fnmotol.cz
Frequently asked questions about genetic testing
Will I pay for the genetic test?
The examination is usually fully covered by health insurance.
Where can I get this test?
In the Czech Republic, this genetic testing is provided by a clinical geneticist. In FNM, the Department of Clinical Genetics is a part of the Institute of Biology and Medical Genetics of the 2nd Medical Faculty of Charles University and FNM (ÚBLG). Your blood will be taken on the day of the examination. The genetic department sends the material for laboratory testing to one of the laboratories that performs this test. When choosing a laboratory, the clinical geneticist considers several things, such as whether the family has already been examined somewhere, the specific experience of the laboratory with ADPKD, and other factors.
The advantage of ÚBLG is that if commonly available tests (gene testing PKD1, PKD2) does not reveal the desired mutation, we can provide research through testing of other commonly unexamined genes associated with kidney disease.
I am the only one in the family with ADPKD, how is this possible when it is an inherited disease?
The mutation could have arisen anew (you are the first carrier and there is a risk of transmission to your offspring). There may also be an incorrectly assigned diagnosis or insufficient examination of relatives. Genetic testing will help clarify the situation.
What does genetic testing entail? How long does the examination take?
Part of the genetic examination is a consultation with a clinical geneticist, who will acquaint you with genetic testing and examine you clinically. This test can detect signs typical of a mutation in a particular gene and speed up otherwise very long laboratory testing. The consultation may also reveal other health problems in you or your family members for whom it would be appropriate to consider genetic testing (for example, testing for oncological predispositions, infertility, thrombophilic conditions, etc.).
The test itself is performed from a single test tube of blood, which is usually taken on the same day.
The laboratory test of the most common mutations takes about 3-4 months. However, it is possible that relatives may need to be tested or the test may not detect a mutation in commonly tested genes, and testing may be significantly prolonged. Therefore, when planning a pregnancy and considering artificial insemination, we recommend that the test be performed as soon as possible.
I have had a genetic test in the past, does it need to be repeated?
It depends on what specific test was performed and what the result was. The latest methods allow us to capture not only a gene but also a specific mutation. Thus, several-year-old tests may no longer be sufficient. In case of any ambiguity, it is possible to contact the attending geneticist or send a question directly to MUDr. Michaela Zelinová (michaela.zelinova@fnmotol.cz).
3D scanning research project
ÚBLG FNM has a facial 3D scanner available for diagnostic and scientific purposes. Our department is currently recruiting patients for a study that deals with the evaluation of the face of patients with a proven pathogenic variant in the genes. PKD1 a PKD2 using facial 3D morphometry. The study is supported by the Grant Agency of Charles University.
Participation in the study is completely voluntary and genetic testing itself is of course possible without participation in this study.
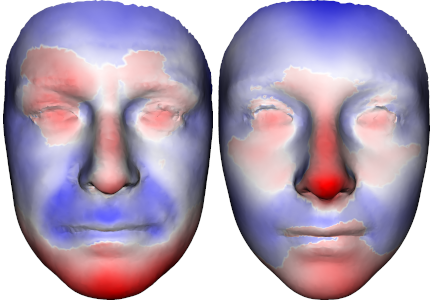
In case you would take part in the study, it is a very fast taking of a picture using a 3D scanner in the premises where there is also a genetic consultation on the day of your examination or communication of results (maximum 5 minutes). The data from the 3D scanner itself will not be evaluated separately but within a large cohort (about 350 patients with ADPKD). Only a summary image of the "average" face of patients with ADPKD will be published (see figure). We consider patients' personal data to be confidential and their protection is our highest priority.
The reward for the participants can be to provide a 3D image of the face (possibility to send your 3D image and the necessary browser by email) and a good feeling that you will help with scientific knowledge and reveal new knowledge about ADPKD.
In the event that a family who has already had a genetic test is willing to participate in the study (a short visit to take a picture), we will be very grateful. According to your time possibilities, we will arrange a short visit to FNM in order to take a 3D image. You can contact MUDr. Michaela Zelinová (michaela.zelinova@fnmotol.cz).
Figure: Visualization of "average" faces of male patients with ADPKD (left) and female patients with ADPKD. The color distinguishes where "something extra" is present compared to the average face of a healthy control (ie without ADPKD) (red), or where, on the contrary, it is missing (blue).
For the professional public
The research is closely related to the diagnostic activities of our institute. The main focus of research is mainly various aspects of human inherited diseases. We mainly deal with a detailed analysis of their molecular nature, but we are not interested in studying their clinical manifestations, population incidence or psychological impacts. Several projects solved at the institute are also focused on somatic mutations in sporadic, ie non-hereditary forms of cancer. The research projects of our institute are interconnected and are connected to research at other collaborating workplaces, both within the 2nd Medical Faculty of Charles University and the Motol University Hospital, and elsewhere in the Czech Republic or abroad. Nevertheless, the projects can be roughly divided into several large areas:
- Cystic fibrosis
- Cytogenetics (microdeletion syndromes, chromosome marker, subtelomeric rearrangements)
- Neurogenetics (muscular dystrophy, fragile X syndrome, FXTAS, spinal cerebellar ataxia, spinal muscular atrophy, Prader-Willi / Angelman syndromes)
- Oncogenetics (hereditary non-polyposis colon cancer (HNPCC), von Hippel-Lindau disease, Li-Fraumeni syndrome, somatic genetic changes in kidney tumors, somatic genetic changes in bladder tumors, somatic mutations of TP53 gene in Barrett's esophagus)
- Psychiatric genetics (autism genetics, ADHD genetics)
- Reproductive genetics
Most of the regular seminars of the institute on Tuesday are devoted to the progress of our research projects (we invite guests to present some interesting topics at some seminars). Postgraduate students are also heavily involved in solving most of our research projects.
For students
Information for students
The Department of Biology and Medical Genetics provides undergraduate and postgraduate teaching of students in the fields of biology and genetics. For undergraduate master's students, ÚBLG provides teaching of the following compulsory subjects in Czech and English:
1st year: Cell, Biology I (Cell, Biology I)
2nd year: Biology II (Biology II)
Teaching compulsory subjects in bachelor's studies:
Biology for the 1st year in the fields of Physiotherapy, General Nursing, Pediatric Nursing
4th year - Clinical genetics
For master's studies, the department also offers the following compulsory elective courses:
Teaching in the Czech language:
Examination methods in medical genetics - teacher RNDr. Eduard Kocarek, PhD.
Geographical medicine and balneography - teacher RNDr. Eduard Kocarek, PhD.
Advances in Molecular Genetics - Teacher Prof. Ing. Zdenek Sedlacek, DrSc.
Reproductive medicine and reproductive genetics - teacher Doc. MD Milan Macek, CSc.
Preconception, preimplantation and prenatal diagnosis, prevention and treatment of severe disorders of prenatal development within the framework of fetal medicine using the latest methods of molecular genetics and stem cells - teacher Doc. MD Milan Macek, CSc.
Teaching in English:
Reproductive Genetics and Reproductive Medicine - teachers Assoc. MUDr. Milan Macek, CSc.
Advances an molecular genetics - teacher Prof. Ing. Zdenek Sedlacek, DrSc.
Examination methods in Medical Genetics - teacher RNDr. Eduard Kocarek, PhD.
CLINICAL GENETICS
- Indications and principles of molecular genetic testing (article by Prof. E. Seemanová, MD, DrSc.) (3,81 MB)
- Chromosomal instability syndromes (article by Prof. E. Seemanová, MD, DrSc.) (2,79 MB)
- When a general pediatrician should send a child for a genetic examination (article by Prof. MUDr. E. Seemanová, DrSc.) (2,64 MB)
- Syndromes with genomic imprinting (article by Prof. E. Seemanová, MD, DrSc.) (5,27 MB)
- Microdeletion syndromes (article by Prof. E. Seemanová, MD, DrSc.) (5,91 MB)
- Genetic syndromology (article by Prof. MUDr. E. Seemanová, DrSc.) (3,01 MB)
- Syndromes and diseases from trinucleotide multiplication mutations (article by Prof. E. Seemanová, MD, DrSc.) (4,21 MB)
- Mosaic phenotypes (article by Prof. MUDr. E. Seemanová, DrSc.) (4,00 MB)
- Significance of chromosomal instability syndromes (Seemanová E., Seeman P., Jarolím P.) (4,71 MB)
TEACHING PROCEEDINGS
- Selected aspects of medical genetics (7,99 MB)
National Coordinating Center for Rare Diseases
In 2012, the "National Coordination Center for Rare Diseases" was established at the ÚBLG and the "Highly Specialized Health Care Center for Cystic Fibrosis Patients" (this center includes the ÚBLG and the Pediatric Clinic) see MZČR Bulletin 4/2012.
The project is supported by a grant from Norwegian funds.
 |  |
Recommendation
As part of the activities of the National Coordination Center for Rare Diseases, it was established in cooperation with Medical Genetics Society ČLS JEP also has the following recommendations:
- Genetic laboratory examination in reproductive genetics
- Recommendations for preimplantation genetic laboratory examination
- Recommendations on laboratory screening of VVV in I. and II. trimester
- Validation of ESHG recommendations for genetic laboratory testing in asymptomatic minors
News
In days September 14 - 16, 2015 took place in connection with the previous congress at the ILF Hotel in Prague professional course again on the topic of rare diseases. The partner of this event is the University Hospital in Motol, Department of Biology and Medical Genetics, holder of the PDP 3 project. More information on the website Czech Internal Medicine Society.
In days 24.-25. September 2015 A conference was held at the Mendel Museum in Brno in cooperation with the National Coordination Center for Rare Diseases and with the support of the project NF-CZ11-PDP-3-003-2014 Human Genetics from Mendel to the Present Day. The main topics were rare diseases, new methods of molecular genetic diagnostics, care of patients with selected rare diseases and ethical aspects. The conference was also attended by the Deputy Norwegian Ambassador Monica Stensland. For more detailed information, see conference program, is also available photo gallery. You can also find more information in the article on the web eeagrants.cz.
On October 19, 2015 The Deputy Norwegian Foreign Minister Elsbeth Tronstad, accompanied by the Norwegian Ambassador to the Czech Republic Siri E. Sletner, visited the University Hospital in Motol. The Norwegian delegation was presented with the current status of the project. During the visit, the technology for obtaining a 3D model of dysmorphic patients was demonstrated, which was purchased from Norwegian funds. The deputy expressed her satisfaction and appreciation for the course of the project so far. More information about visiting Prague can be found at Norwegian Embassy website or Facebook.
October 24, 2015 took place with the support of the project National Coordination Center for Rare Diseases at the University Hospital in Motol Reg. No. NF-CZ11-PDP-3-003-2014 conference Pediatric Dermatology in which MUDr. Hana Bučková, project specialist.
With the support of the project National Coordination Center for Rare Diseases at the University Hospital in Motol, reg. No. NF-CZ11-PDP-3-003-2014, the project experts actively participated in the ČAVO meeting, which took place October 24, 2015 in Prague.
With the support of the project, the following publications were also created, in which the project staff participated:
- “Cost-of-illness analysis and regression modeling in cystic fibrosis: a retrospective prevalence-based study. Authors: Tomáš Mlčoch, Jiří Klimeš, Libor Fila, Věra Vávrová, Veronika Skalická, Marek Turnovec, Veronika Krulišová, Jitka Jirčíková, Dana Zemková, Klára Vilímovská Dědečková, Alena Bílková, Vladimíra Frühaufová, Lukáš Homola, Zuzana Friedmannová, Radovan Drnek, Pavel Dřevínek , Tomáš Doležal, Milan Macek, Jr .. European Journal of Health Economics. Available online.
- Fanconi anemia, complementation group D1 due to biallelic mutation of the BRCA2 gene - case report. Puchmajerova A., Švojgr K., Novotna D., Machačkova E., Sumerauer D., Smišek P., Kodet R., Kynčl M., Křepelova A., Foretova L. Klin Onkol 2016; 29 (Suppl 1): S89 – S 92
- Hepatoblastoma, etiology, case reports. Puchmajerova A., Křepelova A., Indrakova J., Sitkova R., Balaščak I., Kruseova J., Švojgr K., Kodet R., Kynčl M., Vicha A., Macek M. Jr .. Klin Onkol 2016; 29 (Suppl 1): S1 – S 5
On January 22, 2016 the Norwegian partner of the project, Dr. Gunnara Houge of Haukeland University Hospital Bergen (see program below).
The February 29, 2016 is traditionally referred to as Rare Diseases Day. See for more information press release. Several events take place this year as well:
- Day of rare diseases: patient situation and possible solutions
Round table organized by the Health Care Committee of the Chamber of Deputies of the Parliament of the Czech Republic, under the auspices of Vice-Chairman MUDr. David Kasal and in cooperation with the Czech Association for Rare Diseases (from 13.30, PSP CR, Malostranské náměstí 7/19, Prague 1). - Make the voice of rare diseases easier to hear
Meeting at the Mendel Museum in Brno - information about the project of the Veronica Hostěnín Center, concert of the Husák Quartet (from 15.00 pm, Mendel Museum, Mendel Square 1a, Brno). - With his head in the clouds - social evening organized by the Czech Association for Rare Diseases, from 18.30 pm, Žižkov Tower, Mahlerovy sady 1, Prague 3
In the period from 1. 3. 2016 do 30. 6. 2016 Two foreign publications related to the solution of the project NF-CZ11-PDP-3-003-2014 were published:
- Svojgr K, Sumerauer D, Puchmajerova A, Vicha A, Hrusak O, Michalova K, Malis J, Smisek P, Kyncl M, Novotna D, Machackova E, Jencik J, Pycha K, Vaculik M, Kodet R, Stary J. Fanconi anemia with biallelic FANCD1 / BRCA2 mutations - Case report of a family with three affected children. Eur J Med Genet. 2016 Mar; 59 (3): 152-7. doi: 10.1016 / j.ejmg.2015.11.013. Epub 2015 Dec 2. PubMed PMID: 26657402.
- Kouz K, Lissewski C, Spranger S, Mitter D, Riess A, Lopez-Gonzalez V, Lüttgen S, Aydin H, von Deimling F, Evers C, Hahn A, Hempel M, Issa U, Kahlert AK, Lieb A, Villavicencio- Lorini P, Ballesta-Martinez MJ, Nampoothiri S, Ovens-Raeder A, Puchmajerová A, Satanovskij R, Seidel H, Unkelbach S, Zabel B, Kutsche K, Zenker M. Genotype and phenotype in patients with Noonan syndrome and a RIT1 mutation. Genet Med. 2016 Apr 21. doi: 10.1038 / gim.2016.32. [Epub ahead of print] PubMed PMID: 27101134.
Other activities within the monitoring period 1 March 3 - 2016 June 30:
- Autism TV Show - http://www.ceskatelevize.cz/ivysilani/10101491767-studio-ct24/216411058060401
- The future from a drop of blood; Lidové noviny May 4, 2016, supplement Medicine.
- The test also prevents the unexpected death of a child; Lidové noviny May 4, 2016, supplement Medicine.
- Rare diseases: He has the point of approaching solving problems together. Tempus Medicorum, No. 3/2016, volume 25.
- 450 orphan drugs are under development. Medical review 5-6 / 2016.
- Rare diseases in the European context; Milan Macek. May 15, 2016 - 25th Annual Congress of the Society of Cardiology in Brno. Lecture
- Hearing impairment; Radka Pourová. March 29, 3 Pardubice. Lecture
- Invitation to the 3rd National Conference: Care for patients with DMD / BMD
- Standards of care for pediatric DMD patients in the Czech Republic; Jana Haberlová. 3. National Conference: Care of patients with DMD / BMD; April 14, 4, Prague.
- Invitation to the opening of the exhibition "We live with you", held on 15 April 4 in the Atrium of the Motol University Hospital.
- XIII. Annual conference DEBRA ČR, z.ú., held on 8 - 10 April 4 in Brno.
- Introduction to genetics in relation to multiple sclerosis; Eduard Kočárek, 4th year of the multidisciplinary professional conference RS 2016, held on 20 - 21 May 5 in Prague.
- Genetics of autism - approaches / possibilities of examination of patients with PAS; Markéta Havlovicová, lecture. Conference on International Autism Day: "Childhood with Autism" - organized by the association MIKASA zs, on April 1, 4, Faculty of Medicine, University of Ostrava.
- Hepatoblastoma: etiology and selected case reports, Alena Puchmajerová, poster, Conference on familiar cancer, Madrid, 19. -20. May 5
- Hereditary breast and ovarian cancer syndrome, extended testing criteria, news in the care of BRCA gene mutation carriers, Alena Puchmajerová, lecture, Motolské dny 31. 3. -1. April 4
- Rare diseases and molecular genetic diagnostics: Basic overview for general practitioners, lectures, April 30, 2016, Prague, Czech Medical Chamber, Training course
a. Rare hereditary national syndromes; A. Puchmajerová. lecture
- ČAVO zpravodaj 1/16 - articles "Beyond borders or rare diseases elsewhere in Europe", "From the history of rare diseases"
- Affidavit - Number of samples in the biobank
- Intraoral scanner test display
Project activities for the 6th monitoring period
- Brno Genetic Day 2016. Support and active presentation.
- a. Lecture - The Road to the Autistic Mind - MUDr. Markéta Havlovicová. Brno Oncogenetic Day 2016 - 6.10.2016 Brno.
- Conference and meeting of ČAVO members October 15, 2016, Prague. Support and active presentations.
- Lecture - Genetic laboratory examination in rare diseases - MUDr. Marek Turnovec. Conference and meeting of ČAVO members October 15, 2016, Prague.
- Lecture - Student education in cooperation with ČAVO - MUDr. Markéta Havlovicová. Conference and meeting of ČAVO members October 15, 2016, Prague.
- Pediatric Dermatology Conference, October 21.10.2016, XNUMX, Prague. Support and active presentations.
- Lecture - MUDr. Hana Bučková - 15 years of experience in caring for patients with VO
- Rare diseases are differentiated into eight thousand, the general practitioner can hardly detect them. Czech Television 10.9.2016/24/1903427 - www.ceskatelevize.cz/ctXNUMX/domaci/XNUMX-vzacnych-chorob-se-rozlisuje-na-osm-tisic-prakticky-lekar-je-stezi-odhali
- Problem with the diagnosis of rare diseases. Czech Television 10.9.2016/1097181328/216411000100910; www.ceskatelevize.cz/ivysilani/492049-udalosti/XNUMX/obsah/XNUMX-problem-s-diagnozou-u-vzacnych-onemocneni
- Article "Rare experience: Frambu". 3rd year student Markéta Adamovičová. Summer internship 2016. Available at: www.lf2.cuni.cz/fakulta/clanky/reportaze/vzacna-zkusenost-frambu
- Minimum Information required for a DMET Experiment reporting. Kumuthini J, Mbiyavanga M, Chimusa ER, Pathak J, Somervuo P, Van Schaik RH, Dolzan V, Mizzi C, Kalideen K, Ramesar RS, Macek M, Patrinos GP, Squassina A. PMID: 27548815; DOI: 10.2217 / pgs-2016-0015
- A product if immunoreactive trypsinogen and pancreatitis-associated protein as second-tier strategy in cystic fibrosis newborn screening. S Weidler et al. J Cyst Fibros. 2016 Jul 22; DOI: 10.1016 / j.jcf.2016.07.002
- Odontogenetic keratocyst in the Basal cell Nevus (Gorlin-Goltz) Syndrome associated with parethesia of the Lower jaw: case report, reprospective analysis of a representative Czech cohort and recommendations for an early diagnosis of the disease; Milan Hubáček, Tereza Kripnerová, Michaela Němčíková, Anna Křepelová, Alena Puchmajerová, Marcela Malíková, Markéta Havlovicová, Jana Čadová, Roman Kodet, Milan Macek Jr., Tatjana Dostálová; Neuroendocrinol Lett 2016; 37 (4): 101–108
- Human Epididymis Protein 4: A Novel Serum Inflammatory Biomarker in cystic fibrosis. Nagy B Jr, Nagy B, Fila L, Clarke LA, Gönczy F, Bede O, Nagy D, Újhelyi R, Szabó Á, Anghelyi A, Major M, Bene Z, Fejes Z, Antal-Szalmás P, Bhattoa HP, Balla G , Kappelmayer J, Amaral MD, Macek M Jr, Balogh I. Chest. 2016 Sep; 150 (3): 661-72. doi: 10.1016 / j.chest.2016.04.006
- A European Spectrum of Pharmacogenomics Biomarkers: Implications for Clinical Pharmacogenomics. Mizzi C, Dalabira E, Kumuthini J, Dzimiri N, Balogh I, Başak N, Böhm R, Borg J, Borgiani P, Bozina N, Bruckmueller H, Burzynska B, Carracedo A, Cascorbi I, Deltas C, Dolzan V, Fenech A , Grech G, Kasiulevicius V, Kádaši Ľ, Kučinskas V, Khusnutdinova E, Loukas YL, Macek M Jr, Makukh H, Mathijssen R, Mitropoulos K, Mitropoulou C, Novelli G, Papantoni I, Pavlovic S, Saglio G, Setric J, Stojiljkovic M, Stubbs AP, Squassina A, Torres M, Turnovec M, van Schaik RH, Voskarides K, Wakil SM, Werk A, Del Zompo M, Zukic B, Katsila T, Lee MT, Motsinger-Rief A, Mc Leod HL, van der Spek PJ, Patrinos GP. PLoS One. 2016 Sep 16; 11 (9): e0162866. doi: 10.1371 / journal.pone.0162866
- Poster - Facial analysis in patients with hemifacial microsomy by 3D geometric morphometry methods: Poláčková P.1,2, Koťová M., Urbanová W., Moslerová V.
- Presentation Congenital malformations - rare diseases - 1: 2000 growth control. Prof. MUDr. T. Dostálová.
- Anthropomorphometric measurements of students of dentistry and general medicine. Prof. MUDr. T. Dostálová
- APO Summer School II - meeting of the professional public with patients and their patient organizations. August 30, 2016 Milan Macek
- Letter to the Dean of the Faculty of Medicine, Masaryk University in Brno - extension of teaching medical genetics to medical students in cooperation with ČAVO
- Affidavit of the biobank
On Tuesday 28. 2. 2017 The final conference of the project NF-CZ24-PDP-6-8.30-16.30 took place in the Orea Hotel Pyramida (Bělohorská 11, Prague 3) from 003 to 2014. More details in program a press release.
The last day of February is traditionally remembered internationally as a day of rare diseases. This year, this day fell on Tuesday, February 28, 2017. This year's motto "Research gives us hope" refers to the great importance of research for patients with rare diseases. For more information, see, for example, the press release of the Czech Association for Rare Diseases (see appendix) and its website (vzacna-onemocneni.cz).
More information on the page: http://vzacna-onemocneni.cz/ a http://nkcvo.cz/
About the project
The objectives of this project fully correspond to the issues in Program Area No. 27 within the “Public Health Initiatives” (2009-2014) - ie to ensure basic aspects of public health in the field of VO in the Czech Republic in the context of primary, secondary and tertiary VO prevention.
The NKCVO was commissioned within the NS and NAP in cooperation with the OZS MZČR and CAVO to solve them:
- Preparation of the concept of diagnostics and medical-preventive care for VOs based on Norwegian experience and generally accepted European principles (eg based on the recommendations of EUCERD.eu, Eurordis.org), adapted to current medical and preventive care in the Czech Republic and preparation of expert documents for audits of existing VO centers and their further work in accordance with the proposed concept in point 1), including professional documents for the methodology of establishing new centers in the health care system in accordance with §112 of Act 372/2011 Coll. OZS MZ ČR.
- To increase awareness and knowledge in VO issues among the lay and professional public (doctors, non-doctors and other medical staff) and in undergraduate and postgraduate teaching in cooperation and close links and exchange of results with Norwegian partners (min. 3 training events, Chapter 1 in monograph, 1st special issue of the journal). A partial goal is the development of information portals for VO.
- Improve detection and secondary prevention in genetically at risk families. NKCVO will introduce and improve the algorithm of genetic testing in genetically risky families with VO, within the framework of specialized genetic counseling at the level of all departments of medical genetics (state and non-state health care facilities). Another partial goal is to ensure the most accurate "digital" phenotyping of VO, whether in terms of anthropological, clinical and genealogical with the indication of specific laboratory / molecular genetic and cytogenetic examinations and follow-up to syndromological diagnostics (output: phenotypic examination algorithm) and to develop a neuropsychiatric examination algorithm evaluation of the degree of psychosomatic disability and threat to normal development in children and adults with VO (output: examination algorithm). At the same time, to ensure at the level of specialized workplaces the optimal combination and standard of molecular cytogenetic methods (arrayCGH, MLPA, NGS) to ensure preimplantation, prenatal and postnatal diagnosis of genetically precisely defined syndromes accompanying VO in close connection and exchange of results with Norwegian partners (output: methodology for genetic sites). Described in the activity of elaboration of recommended procedures for ensuring clinical-genetic and molecular genetic diagnostics of VO.
- Carry out model pharmacoeconomic studies at selected VOs for objectifying reimbursement of treatment procedures at VOs in cooperation with SUKL Prague and iheta.org.
- Continuous expansion of the frozen sera biobank to further improve diagnostics in families with VO in the FNM cryocenter. Described in the activity of elaboration of recommended procedures for ensuring clinical-genetic and molecular genetic diagnostics of VO.
- Organization of the final international conference with presentation of project outputs.
As of 3 August 2015, an additional project activity was approved, the purpose of which is to create a database of genomic variants in a representative cohort of the Czech population on the basis of whole-genome sequencing of min. 200 individuals with the help of a pipetting system, which will be acquired within the project.
As part of this additional activity, a pilot study will be carried out in which standards of body composition, proportions and body composition of preschool children in categories 4, 5 and 6 will be developed for 30 people of each sex. 13 dimensions will be measured: body height, weight, head circumference, length and width of the skull, width and height of the face, sitting height, upper limb span, walking length, arm circumference, skin line above the triceps and above the scapula. The sub-activity includes the development of developmental standards for the face, floor and evaluation software. Facial development standards will be developed in the form of average virtual surface models (including variability) from 0 to 15 years, optimally from at least 30 - 50 individuals for each year of age and sex, ie 1000 - 1600 models, including adjustment and further analytical processing. Morphome3cs software will be modified: user interface, export of DCA outputs for calculation of standards on surface models, average shapes for selected subgroups, statistical model for face shape for individual sexes, PCA score, topological information, import of individual surface model (OBJ format), manual inserting landmarks on a surface model, import of pre-prepared standards, comparison of an individual against a standard, map of surface distances, Mahalanobis distance of main components in space, other statistics. The Faculty of Science of Charles University in Prague will take part in this pilot project.
In cooperation with the Dental Clinic of Charles University 2nd Medical Faculty and the University Hospital in Motol, another partial project activity will be solved, which is focused on upper floor examination standards in the form of average virtual models for 4 age categories: 5 years (10 x), 12 years x), 20 years (15 x), 20 years (18 x). An intraoral 20D scanner will be used for this processing, which will be purchased from the project.
The project reg. No. NF-CZ11-PDP-3-003-2014 entitled National Coordination Center for Rare Diseases at the University Hospital in Motol until April 30, 2017. The total cost of the project is CZK 30, of which the subsidy from Norwegian funds is 972% ie CZK 417 and 80%, ie CZK 24, is co-financing.
Project schedule
NKCVO - project schedule
- Project schedule (49,96 KB)
- Project schedule (62,45 KB)
Project description
Rare diseases (hereinafter VO) are heterogeneous, multisystem diseases with a significant impact on the quality of life and social inclusion of patients. In accordance with the "EC 141/2000" regulation, VOs affect <5 per 10.000 people. Although VOs are individually rare, there are more than 8000 diseases, of which only 200 are in ICD10 (www.orpha.net), so the total number of patients in the Czech Republic is approx. 20. VOs often occur after birth, and may affect 000-4% of newborns and infants (eg metabolic disorders, genetic syndromes and rare tumors), but may also occur in adulthood. Approximately 5% of VOs are due to highly penetrant mutations, the remaining VOs are multifactorial or due to epigenetic environmental influences, often their pathogenesis is unknown. Incorrect or late diagnosis leads to irreversible damage to health, psychological trauma to patients and their families, including distrust of the quality of health care. The detection and clinical diagnosis of VO depends primarily on the training of general practitioners.
The tasks of the project can be summarized in four "4P" points: a) the need to "think" about the possibility of VO due to the primary clinical suspicion in field practice; b) VO "know" - ie correctly diagnose in specialized centers; c) "help" - ie ensure their effective prevention and treatment; and d) "finance" and prepare pharmacoeconomic data for the introduction of medicinal products for VO.
The tasks of the "4P" project are reflected in the project activities, thus ensuring the objectives of the approved Program Agreement (PA) CZ11 - Annex I - point 6.3 and the project outputs will be used even after the end of the project. The project requires financial support for the development of the activities of the National Coordination Center for VO (NKCVO) at the ÚBLG in the Motol University Hospital and the 2nd Medical Faculty of Charles University in Prague. NKVCO will cooperate with OZS MZČR and the Czech Association for Rare Diseases (ČAVO), which is a member of the European association Eurodis.org. The required funding from Norway Grants will enable the operation of the NCCVO, which is equipped with infrastructure from previous domestic and international grant activities.
The planned activities of the NKCVO have the task in the sense of "4P" and in accordance with the tasks of the NAP:
- to expand awareness and knowledge of VO issues among the professional and lay public in cooperation with ČAVO and the Norwegian partner Frambu.no within the framework of population, primary VO prevention. Activity - raising awareness among the lay and professional public VO
- The NCCVO will cooperate with the OZS MZČR within the activities of the "Interdepartmental and Interdisciplinary Commission for VO" on the development of existing activities in preparation for the establishment of new centers for the improvement and standardization of medical care for VO. Activity - preparation of professional documents for public health projects related to VO
- introduction of the latest methods of molecular cytogenetic and genetic diagnostics with a special focus on the latest new generation sequencing (NGS) methods for prenatal and postnatal diagnosis of VO and clarification of the molecular pathogenesis of hitherto uncharacterized VO, including their prospective treatment.
- 4) improving the pharmacoeconomic estimation of diagnostic and treatment costs for selected VOs in cooperation with SÚKL. Activity - preparation of professional documents for public health projects related to VO
- improving comprehensive care for VO by validating and using the experience of Norwegian partners in the field of public health, incl. application of experience Frambu and partner org. ČAVO. Activity - elaboration of recommended procedures for ensuring clinical-genetic and molecular genetic diagnostics of VO
- development of undergraduate VO teaching for medical students and postgraduate specialized training of doctors within the 1st and 2nd Faculty of Medicine, Charles University and IPVZ.
International cooperation
The cooperation of two foreign partners is ensured within the project. One of them is the Center for Medical Genetics and Molecular Medicine, Haukeland University Hospital, Bergen is a leading Norwegian academic medical institution with which we will work in improving NGS clinical-genetic methods, syndromological examination, detection of inherited metabolic disorders, and development of molecular cytogenetic and genetic methods of early prenatal diagnostics, bioinformatics and digital phenotyping of VO. The NKCVO already has a long-term cooperation with the partner within the activities of the European Society of Medical Genetics (www.eshg.org) and the cooperation will continue during the sustainability of the project.
The second foreign partner is Frambu.no - "Norwegian Resource Center for Rare Disorders" is the main government expert agency for VO, which will cooperate with NKCVO and ČAVO in raising awareness of the professional and lay public about VO to improve the detection of genetically at risk families. Cooperation with Frambu will be newly created within this project and will continue during the sustainability of the project.
For more information on the cooperation with FRAMBU and the Norwegian Institute of Public Health, see this article in anchlitina whether Norwegian.
Contact
Prof. MD Milan Macek, DrSc., MHA - Main project coordinator
Phone: + 420 224 433 501
Fax: +420 224 433 520
Email: milan.macek.jr@lfmotol.cuni.cz
Ing. Ivana Funková, MBA - Financial and administrative support of the project
Phone: + 420 224 433 501
Fax: +420 224 433 520
Email: ivana.funkova@fnmotol.cz
Ing. Vera Mozrová - Financial and administrative support of the project
Phone: + 420 224 431 141
Email: vera.mozrova@fnmotol.cz
ACGT project
Analysis of Czech genomes for theranostics
FN Motol has joined the ACGT project and is happy to welcome newcomers.
Are you interested in participating in one of the largest genetic projects?
Want to find out where your ancient ancestor probably came from?
PROJECT GOAL:
Compilation of a control database of genetic information typical of the Czech population based on the analysis of genetic information of more than 1 volunteers.
USE PROJECT RESULTS:
- Improving the diagnosis and therapy of genetic diseases
- Research into a number of genetic diseases
PROJECT COURSE
- Recruitment of 1500 volunteers in 2019-2022
- Performing a genographic analysis on each of the volunteers
- Determining the sequence of their genome - all genetic information
- Compilation of a control Czech genetic database
WHAT IS GENOM?
The genome is a collection of all the genetic information found in DNA. It is present in almost every cell in you and contains instructions important for the formation and functioning of the body. He is responsible for inheriting traits from parents to descendants. It contains 3,2 billion base pairs, which you can think of as two rows of 3,2 billion differently arranged letters A, C, G and T.
WHAT IS SEQUENCE AND WHY IS IT CARRIED OUT?
When sequencing the genome, the order of all 3,2 billion letters in your DNA will be read. In this way, we will read the genome of at least one thousand Czechs and compile a database that will show how many people have a letter in a given position. This will help determine whether some variants (letter substitutions) in patients' DNA are rare or whether they are common for the Czech population.
WHAT IS TERANOSTIKA?
Teranostika is a compound of the words THERAPY and DIAGNOSIS. This is a new term from the field of personalized medicine. She prepares tailor-made treatment for each patient, according to his needs. Each person is judged individually, because he is unique in terms of genetic predisposition, and therefore his reaction to the treatment procedure may also differ. Knowledge of the genome typical of the Czech population will enable significant progress in this sector.
CONDITIONS FOR PARTICIPATION IN THE PROJECT
- Age 30-55 years
- The participant's parents come from the same region (the father was born in the same region as the mother)
- A healthy individual, ie. without a serious genetic disease
- Participation is not possibly in the case of an ongoing pregnancy, receiving a blood transfusion in the last six months or undergoing a transplant at any time in life.
REMUNERATION FOR PARTICIPATION IN THE PROJECT
- Free genographic analysis - according to it, it is possible to estimate where your ancient ancestor probably came from.
- More on www.rekreacnigenetika.cz in the Genographer section.
WHAT TO DO IN CASE OF INTEREST
- On the project website www.ACGT.cz you will get information about the project and a questionnaire, which lists, among other things, the conditions for inclusion in the project.
- You will arrive at the collection point. You either fill in the questionnaire yourself in advance at home (preferred) or with the help of an authorized employee in the transfusion department.
Opportunity to participate in the project at FNM:
Responsible person: Erika Marková
Tel .: +420 224 433 583 (between 9 am and 15 pm, must be ordered in advance on tel. 224 433 583) or E-mail: erika.markova@fnmotol.cz
V Úvalu 84, Prague 5; main blue building, node D, 3rd floor, Genetic Outpatient Clinic
Project website: http://www.acgt.cz/
Health Program - EEA Funds 2014-2021
| Project name: | Increasing the availability of targeted prevention and early diagnosis of specific communicable and non-communicable diseases in selected socially excluded localities with Roma communities |
| Register. number: | ZD-ZDOVA2-001 |
| Allocated funds: | 19.563.645 CZK |
| Supported by a grant from Norway in the amount of: | 16.629.098 CZK |
| Amount of co-financing from the state budget: | 2.934.547 CZK |
| Implementer: | University Hospital in Motol, Department of Biology and Medical Genetics |
| Implementation time: | From the 1st of April 2021 to the 30th April 2024 |
| Professional project guarantor: | prof. MD Milan Macek, DrSc., MHA |
| Project manager: | Ing. Ivana Funkova, MBA, Ing. Věra Mozrová |
| Contact information: | University Hospital in Motol, V Úvalu 84, 150 06 Prague 5 - Motol phone: 00420 22443 3501, e-mail: ivana.funkova@fnmotol.cz |
Project goals
The main goal of the project is to improve the availability of specialized health care for the inhabitants of excluded localities, together with a focus on specific non-communicable diseases and the consequent increased risk of communicable diseases, including child injuries, among the Roma ethnic group. From a societal point of view, this project will not only improve the diagnosis and prevention of these diseases, but also systematically set up primary and specialized care in this hitherto insufficiently analyzed and addressed area of health care in the Czech Republic, ie develop the concept of health care pathways for Roma communities in cooperation with leading foreign collaborators from Norway and Romania. The overall benefit of this project is therefore primarily in the area of improved health care and training of relevant health professionals (field and specialized) and paramedical staff, but also secondarily due to long-term economic savings of public health insurance due to more effective care for members of Roma communities and also due to rationalization. indications for social care by the cooperating Coordinators of the SZU Regional Health Promotion Centers.
The project supports target groups within the excluded localities, which include: a) Roma ethnicity of all ages; b) People at risk of or in poverty from the Roma community or the majority population; c) Public 1) lay - in terms of raising awareness of specific diseases in the Roma population from excluded localities and 2) professional public - in terms of specifics and indication criteria for diseases that are relatively more common in the Roma population - ie, pediatricians and adolescents, pediatric neurologists, ophthalmologists, cardiologists, pediatricians and pediatric injuries.
Abbreviations
Here you will find an overview of abbreviations that may appear in reports from our workplace. The list is continuously updated.
| Abbreviation | Importance |
|---|---|
| 2. LF | 2nd Faculty of Medicine |
| AD | autosomal dominant (type of inheritance) |
| ADHD | attention-deficit / hyperactivity disorder |
| ADPKD | autosomal dominant polycystic kidney disease |
| AK | amino acid |
| ALS | amyotrophic lateral sclerosis |
| AMC | amniocentesis - amniotic fluid collection |
| AR | autosomal recessive (type of inheritance) |
| ARPKD | autosomal recessive polycystic kidney disease |
| AS | Angelmann syndrome |
| AS | Asperger's syndrome |
| AS | Apgar score |
| BAP | bipolar affective disorder |
| BMD | Becker muscular dystrophy |
| BMD | Becker muscular dystrophy |
| BPD | biparietal diameter |
| BRCA | breast carcinoma (breast cancer) |
| BWS | Beckwith-Wiedemann syndrome |
| ca | cancer, cancer |
| CF | cystic fibrosis |
| CMP | stroke |
| CMT | Charcot-Marie-Tooth disease |
| CMV | cytomegalovirus |
| CVS | chorionic villi sampling |
| DM | d |
| DMD | Duchenne muscular dystrophy |
| DMO | cerebral palsy |
| DMP | hereditary metabolic disorder |
| DNA | deoxyribonucleic acid |
| DS | Down's syndrome |
| FA | Fanconi's anemia |
| FA | pharmacological history |
| FAS | fetal alcohol syndrome |
| FNM | Motol University Hospital |
| foa | foramen ovale aperture |
| FSGS | focal segmental glomerulosclerosis |
| FXS | fragile X syndrome |
| GPK | Department of Gynecology and Obstetrics |
| hCG | human chorionic gonadotropin |
| HD | Huntington's disease |
| HP | hereditary pancreatitis |
| HSV | herpes simplex virus |
| IBD | inflammatory bowel disease |
| IM | heart-attack |
| IP | pigmented incontinence |
| my | silent pregnancy |
| MPS | mucopolysaccharidosis |
| MR | magnetic resonance |
| MR | mental retardation |
| MRI | magnetic resonance imaging |
| NF | neurofibromatosis |
| NTD | nerve tube defect |
| OA | personal anamnesis |
| OCD | obsessive-compulsive disorder |
| PAPP-A | pregnancy - associated protein |
| Administrative staff | autism spectrum disorder |
| PCOS | polycystic ovary syndrome |
| PE | preeclampsia |
| PMR | psychomotor retardation |
| PMV | psychomotor development |
| PWS | Prader-Willi syndrome |
| RNA | ribonucleic acid |
| huh | miscarriage |
| SCA | spinocerebellar ataxia |
| SIDS | sudden infant death syndrome |
| SMA | spinal muscular atrophy |
| T13 | trisomy of chromosome 13 |
| T18 | trisomy of chromosome 18 |
| T21 | trisomy of chromosome 21 |
| TBC | tuberculosis |
| TCS | Trecher-Collins syndrome |
| TM | thrombophilic mutations |
| Tx | transplantation |
| uE3 | unconjugated estriol |
| UK | Charles University |
| UPT | abortion |
| UUT | abortion |
| UZ | ultrasound |
| VHL | von Hippel-Linda disease |
| VSV | congenital heart disease |
| Tourist | congenital malformation |
| Vzv | varicella zoster virus |
| WBS | Williams-Beurren syndrome |
| ÚBLG | Department of Biology and Medical Genetics |
Contact
Department of Biology and Medical Genetics, 2nd Faculty of Medicine, Charles University and University Hospital Motol
V Úvalu 84
150 06 Prague 5 - Motol
Patient ordering
- Phone: (+420) 224 433 564 (only on working days 7:15-9:15 and 14:00-15:00)
- See separate page for more information Ambulance.
Secretariat
- Telephone: (+420) 224 433 500, (+420) 224 433 501
- Fax: (+420) 224 433 520
- Website: https://www.fnmotol.cz/ublg/
- Email: ublg@fnmotol.cz
School part of ÚBLG
- Plzeňská 130/221
150 06 Prague 5 - Motol - Phone: (+ 420) 257 296 153